Atila Real-Time PCR RhDGT Kit
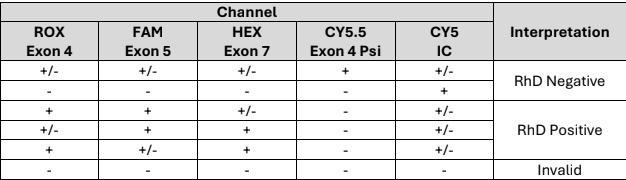
The Atila Rhesus D Genotyping Testing (RhDGT) Kit is for molecular biology research use only and tests for detecting the presence of RhD exon 4, exon 5 and exon 7 on chromosome 1 that indicates RhD positive blood type, or RhD exon 4 pseudogene form with a 38-bp insertion that indicates RhD negative blood type. The kit analyzes the genomic or cell free DNA obtained from the blood and applies the real-time qPCR platform to accurately identify exon 4, exon 4 pseudogene form, exon 5 and exon 7 on chromosome 1.
The Atila RhDGT Kit is intended for use by qualified and trained laboratory personnel specifically instructed and trained in the techniques of real-time nucleic acid amplification.
This product is for research use only (RUO).
Each kit includes 100 tests.
Atila Real-Time PCR RhDGT Kit
The Rh blood group system is one of the most clinically significant blood group systems in transfusion medicine. The presence or absence of the Rhesus D (RhD) antigen on red blood cells (RBCs) determines an individual’s Rh status as either RhD positive or RhD negative. RhD-negative individuals may develop alloantibodies against RhD-positive RBCs after transfusion or pregnancy, which can lead to hemolytic disease of the fetus and newborn or complications in transfusion therapy.
The RhD gene is located on chromosome 1 and consists of multiple exons encoding the RhD protein. Deletions, insertions, and rearrangements in the RhD gene can result in RhD-negative phenotypes. One of the most common causes of RhD negativity is the complete deletion of the RhD gene, while some individuals carry an RhD pseudogene containing a characteristic 38-bp insertion in exon 4, which leads to a non-functional RhD protein.
The Atila Rhesus D Genotyping Testing (RhDGT) Kit is a molecular assay designed to detect the presence of RhD exon 4, exon 5, and exon 7, as well as the exon 4 pseudogene form with a 38-bp insertion. The kit utilizes real-time quantitative PCR (qPCR) to analyze genomic DNA or cell free DNA extracted from whole blood samples, providing a reliable and precise determination of RhD status at the molecular level and includes Internal Control (IC) for monitoring test performance in each individual test. This test is intended for research use only and should be performed by trained laboratory personnel with expertise in molecular biology techniques.
Features Include
Technical Specs
Workflow Overview for Genomic DNA samples.
This section is for demonstrative purposes only and may be incomplete or inaccurate. Always refer to the product instructions for precise guidelines and directions.

Master Mix Preparation

Reaction Setup: 9.5 μL RhDGT primer mix and 7.5 μL RhDGT buffer mix in each tube. Top up the purified genomic DNA or cfDNA sample with H2O to 9 μL

Run on standard PCR instrument