iAMP® Vaginal Panel
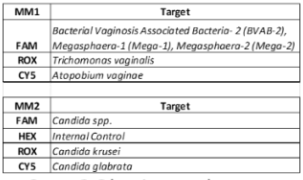
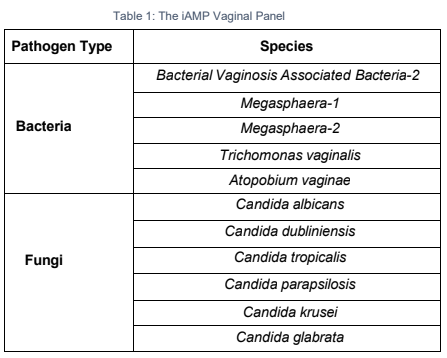
The iAMP ® Vaginal Panel is a real-time fluorescent isothermal PCR assay based on Atila’s proprietary isothermal amplification technology intended for the simultaneous qualitative detection of nucleic acids from 11 pathogens associated with bacterial and fungal infections commonly found in vagina. It can also differentiate Trichomonas vaginalis and Atopobium vaginae from other bacterial infection as well as Candida krusei and Candida glabrata from other Candida species.
Positive results are indicative of the presence of pathogens. Positive results do not rule out co-infection with other pathogens. Negative results do not preclude vaginal infection.
The iAMP ® Vaginal Panel is intended for use by trained laboratory personnel specifically instructed and trained in the techniques of real-time nucleic acid.
RUO in the US.
iAMP® Vaginal Panel
Vaginal infections are among the most common conditions affecting women worldwide and can be caused by a wide range of pathogens, including bacteria, parasites, and fungi. These infections are often associated with symptoms such as abnormal discharge, odor, irritation, and discomfort, and may lead to complications including pelvic inflammatory disease, infertility, and adverse pregnancy outcomes if left untreated. Accurate and timely identification of the causative pathogen is therefore essential for guiding appropriate therapy, preventing recurrence, and reducing the misuse of broad-spectrum antimicrobials. Distinguishing between specific organisms such as Trichomonas vaginalis and Atopobium vaginae from other bacterial infections, as well as identifying antifungal-resistant strains like Candida krusei and Candida glabrata from other Candida species, is critical for improving clinical outcomes.
The iAMP® Vaginal Panel is a high-multiplex molecular diagnostic assay designed to improve the accuracy and efficiency of detecting 11 pathogens associated with vaginal infections. Using advanced multiplex technology, the panel can differentiate Trichomonas vaginalis and Atopobium vaginae from other bacterial pathogens. In addition, the assay identifies clinically significant Candida species, including Candida krusei and Candida glabrata, which are often resistant to standard antifungal medications.
Features Include
Technical Specs
Workflow Overview for the iAMP® Vaginal Panel
This section is for demonstrative purposes only and may be incomplete or inaccurate. Always refer to the product instructions for precise guidelines and directions.
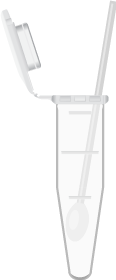
Dry swab in sample tube.

Add 750µL to 1x sample buffer.

Vortex and incubate at 95°C for 10 minutes.
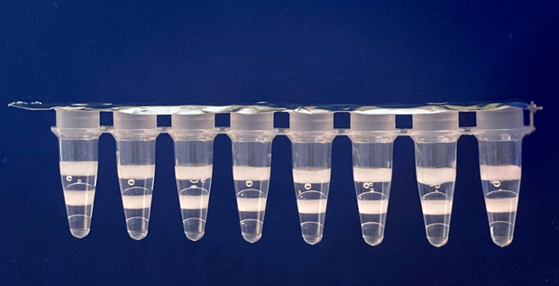
Add 10µL sample to Zebra BioDome tubes.

Run on standard PCR instrument.