Atila HBV/HCV/HIV Assay Kit
The Atila HBV/HCV/HIV Assay Kit is an extraction-free real-time reverse transcription assay test for the in vitro qualitative detection of Human Hepatitis B Virus (HBV), Human Hepatitis C Virus (HCV) and Human Immunodeficiency Virus (HIV-1) in human plasma, serum, and dry blood spot. This test is not intended for use as a screening test for HBV/HCV/HIV or an aid in diagnosis of infection with HBV/HCV/HIV.
The Atila HBV/HCV/HIV Assay Kit is intended for use by qualified and trained laboratory personnel specifically instructed and trained in the techniques of real-time nucleic acid amplification procedures.
This product is RUO.
Each kit comes with 100 tests.
Atila HBV/HCV/HIV Assay Kit
The genome of Hepatitis B virus (HBV) is a partially double-stranded circular DNA of about 3.2 kilobase (kb). HBV infection leads to a wide spectrum of liver diseases ranging from acute hepatitis (including fulminant hepatic failure) to chronic hepatitis, cirrhosis, and hepatocellular carcinoma (HCC). Risk for chronic infection is related to age at infection: about 90% of infants with hepatitis B go on to develop chronic infection, whereas only 2%–6% of people who get hepatitis B as adults become chronically infected. HBV is spread when blood, semen, or other body fluids from a person infected with the virus enters the body of someone who is not infected. This can happen through sexual contact; sharing needles, syringes, or other drug-injection equipment; or from mother to baby at birth.
Hepatitis C virus (HCV) is a single-stranded RNA virus, with a genome size of approximately 9500 nucleotides. HCV can cause Hepatitis C, a liver disease, and is a major cause of liver cancer. The virus causes both acute and chronic hepatitis. Around 30% (15–45%) of infected persons spontaneously clear the virus within 6 months of infection without any treatment. However, the remaining 70% (55–85%) of persons will develop chronic HCV infection. Of those with chronic HCV infection, the risk of cirrhosis ranges between 15% and 30% within 20 years. The HCV is a bloodborne virus, is transmitted by sharing injection equipment, transfusion or exposure to the infected blood, and can also be transmitted sexually or from infected mother to baby.
The human immunodeficiency virus (HIV) belongs to the genus Lentivirus and can cause acquired immunodeficiency syndrome (AIDS). According to World Health Organization (WHO), there are approximately 38 million people living with HIV/AIDS in the world. Two types of HIV have been characterized: HIV-1 and HIV-2. HIV-1 is more virulent and more infectious, and most cases worldwide are caused by HIV-1. HIV-1 genome consists of two identical single-stranded RNA molecules that are approximately 9200-9600 nucleotides long. Both HIV-1 and HIV-2 are transmitted by exposure to contaminated blood or blood and plasma products, exposure to certain body tissues or fluids, by sexual contact or by an infected mother to the newborn child.
The Atila HBV/HCV/HIV Assay Kit is an isothermal nucleic acid reversal transcription amplification assay based on a proprietary technology developed to improve the efficiency of detecting multiple viral nucleic acid targets simultaneously by reducing non-specific interactions and amplifications. DNA oligos are designed to specifically detect and differentiate HBV/HCV/HIV DNA/RNA.
Features Include
Technical Specs
Workflow Overview Plasma Sample
This section is for demonstrative purposes only and may be incomplete or inaccurate. Always refer to the product instructions for precise guidelines and directions.

Obtain plasma sample.
Mix 10uL plasma sample with 10uL 2xSBM.

Vortex briefly (2-3s) and let tubes sit at room temperature for 20-30 minutes.
Add 10µL sample into Zebra BioDome tubes.

Run on standard PCR instrument.
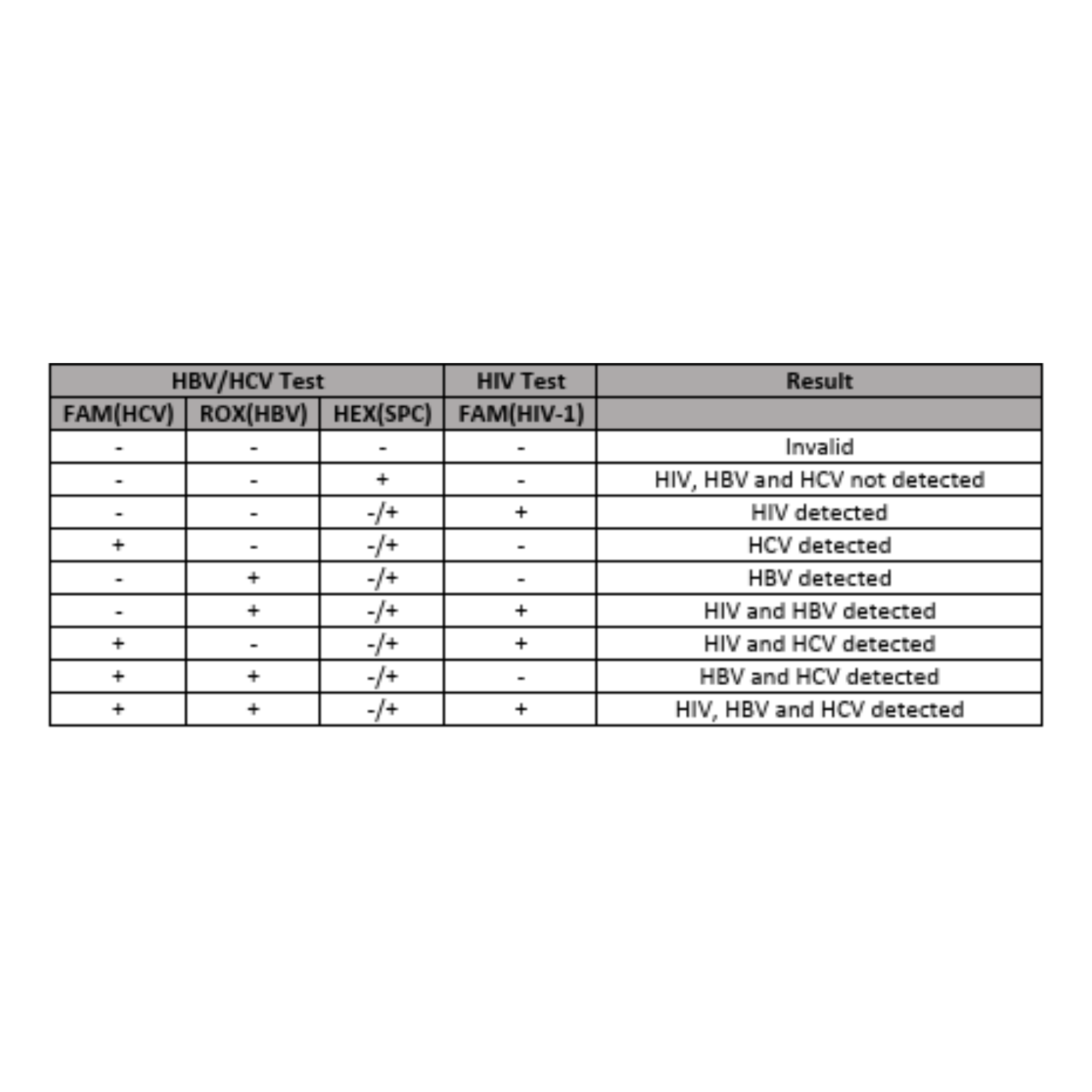
Obtain result report in less than 60 minutes.
Workflow Overview Dry Blood Spot Sample
This section is for demonstrative purposes only and may be incomplete or inaccurate. Always refer to the product instructions for precise guidelines and directions.

Obtain dry blood spot sample and with a sample puncher, punch out sample disk and place in tube.

Add 150uL 1X SBM to tube.

Let tubes sit at room temperature for 10 minutes. Vortex briefly (30s).

Add 10µL sample into Zebra BioDome tubes.

Run on standard PCR instrument.


