Atila BRCA1&2 Mutation Screening Kit
The Atila BRCA1&2 Mutation Screening Kit is a real-time nucleic acid amplification assay for the in vitro qualitative detection of 38 common pathogenic mutations in the BRCA1 and BRCA2 genes. The assay is designed to identify selected mutations associated with an increased risk of hereditary breast and ovarian cancer (HBOC) and related hereditary cancer syndromes. This test can be used to screen human genomic DNA samples to aid in the identification of individuals who may carry clinically relevant BRCA1/2 mutations. It is not intended for general population screening or for use as the sole basis of diagnosis, risk assessment, or clinical decision-making without appropriate clinical correlation, genetic counseling, and confirmatory testing.
This product is for research use only (RUO).
Each kit includes 100 tests.
Atila BRCA1&2 Mutation Screening Kit
Hereditary breast and ovarian cancer (HBOC) syndrome is one of the most common inherited cancer predisposition disorders worldwide and is primarily caused by pathogenic mutations in the BRCA1 and BRCA2 genes, which encode key proteins involved in DNA damage repair through homologous recombination. According to the World Health Organization (WHO), breast cancer is the most frequently diagnosed cancer globally, and inherited BRCA1/2 mutations substantially increase an individual’s lifetime risk of developing breast, ovarian, and other associated cancers. The prevalence of BRCA mutations varies among populations and ethnic groups, with certain recurrent mutations occurring at higher frequencies in specific regions or ancestries. Individuals carrying BRCA1 or BRCA2 mutations are at increased risk of early-onset disease, multiple primary cancers, and aggressive tumor phenotypes, contributing to significant morbidity and mortality. Early and accurate detection of clinically relevant BRCA1/2 mutations is therefore essential for hereditary cancer risk assessment, genetic counseling, personalized surveillance, and informed clinical management strategies.
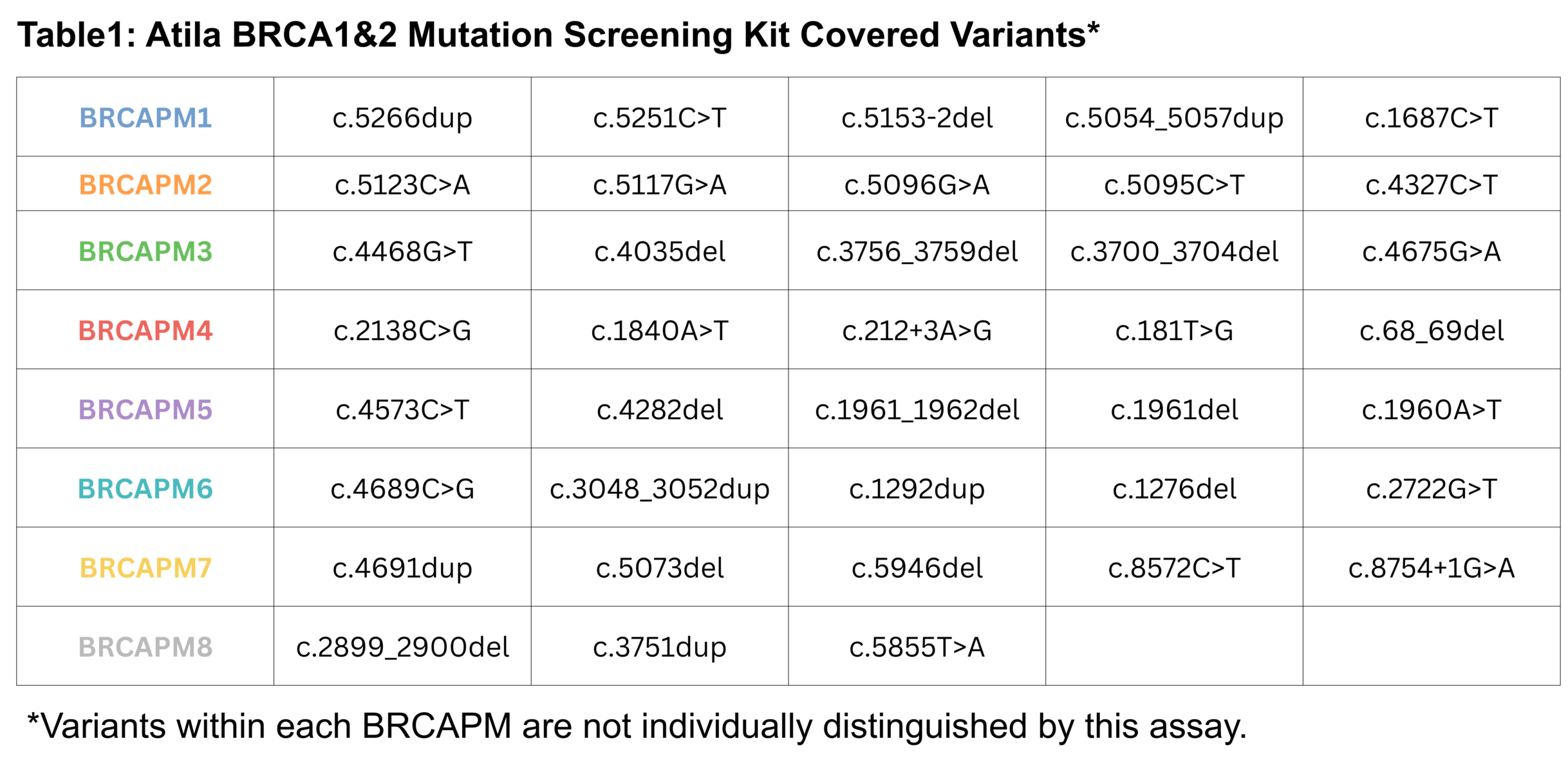
The Atila BRCA1&2 Mutation Screening Kit is a multiplex real-time polymerase chain reaction (PCR) assay based on proprietary technology designed to improve the accuracy and efficiency of detecting selected pathogenic mutations associated with hereditary breast and ovarian cancer syndrome. Multiple sets of oligonucleotides are specifically designed to detect 38 predefined BRCA1 and BRCA2 mutations, which are grouped into several primer mixes (BRCAPMs). The assay detects the presence of a mutation within the coverage of each BRCAPM; however, individual variants that are covered by the same PM are not distinguished. The covered mutations include clinically significant single-nucleotide substitutions, small insertions and deletions, and splice-site variants, as listed in table 1. This test enables reliable screening for common BRCA1/2 mutations to aid in the identification of individuals at increased genetic risk when used in conjunction with clinical evaluation and confirmatory testing.
Features Include
Technical Specs
Workflow Overview for Purified Human DNA.
This section is for demonstrative purposes only and may be incomplete or inaccurate. Always refer to the product instructions for precise guidelines and directions.

Reagent reparation and master mix preparation

Add sample and prepare plate

Run on standard PCR instrument