Atila Sickle Cell Disease Detection Kit (dPCR)
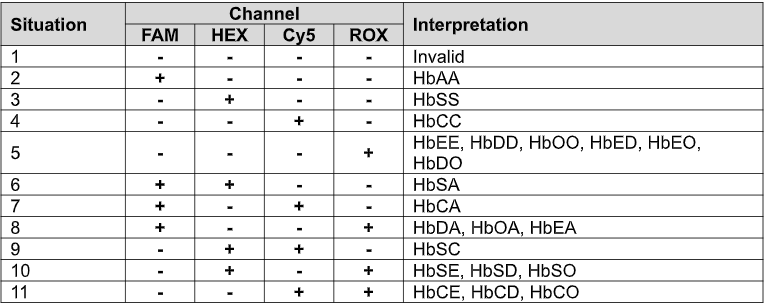
The Atila Sickle Cell Disease Detection Kit is a digital nucleic acid amplification assay (dPCR) for the in vitro qualitative/quantitative detection of mutations associated with sickle cell disease (SCD). The assay is capable of detecting hemoglobin variants HbS, HbC, and HbA individually. Variants HbD, HbO, and HbE can also be detected, although they cannot be distinguished from one another. In addition, the assay can identify compound heterozygous genotypes (e.g., HbSC, HbSE, HbAE). This test can be used to screen for these variants in human blood specimens to aid in the identification of carriers and individuals affected by sickle cell disease or related hemoglobinopathies. It is not intended for general health screening or for use as the sole basis of diagnosis without clinical correlation.
The Atila Sickle Cell Disease Detection Kit is intended for use by qualified laboratory professionals who have been specifically trained in nucleic acid amplification techniques and in vitro diagnostic procedures.
Each kit includes 100 tests.
This product is for research use only.
Atila Sickle Cell Disease Detection Kit (dPCR)
Sickle cell disease (SCD) is one of the most common inherited blood disorders worldwide, caused by mutations in the hemoglobin β-gene that lead to abnormal hemoglobin variants. According to the World Health Organization (WHO), approximately 300,000 infants are born each year with SCD, with the highest prevalence in sub-Saharan Africa, India, the Middle East, and among populations of African descent worldwide. Individuals with SCD are at risk of severe complications, including chronic hemolytic anemia, painful vaso-occlusive crises, stroke, and organ damage, which contributes to significant morbidity and early mortality. Early and accurate detection of hemoglobin variants is therefore essential for disease management, newborn screening, genetic counseling, and transfusion safety. Reliable molecular assays for SCD can improve clinical outcomes by enabling timely diagnosis and guiding appropriate treatment and preventive care strategies.
The Atila Sickle Cell Disease Detection Kit is a multiplex digital polymerase chain reaction (dPCR) assay based on proprietary technology designed to improve the accuracy and efficiency of detecting hemoglobin variants associated with sickle cell disease. Multiple sets of oligonucleotides are specifically designed to detect and discriminate HbS, HbC, and HbA alleles, while also identifying the presence of HbD, HbO, and HbE variants, which cannot be distinguished from one another by this assay. The test is capable of identifying both homozygous and compound heterozygous genotypes (e.g., HbSS, HbSC, HbSE, HbAE), enabling reliable screening of carriers and affected individuals.
Features Include
Technical Specs
Workflow Overview for Purified Genomic DNA (ASCD-100B-RUO)
This section is for demonstrative purposes only and may be incomplete or inaccurate. Always refer to the product instructions for precise guidelines and directions.

Digital PCR assay setup.

Prepare ddPCR Multiplex Supermix, ddPCR Supermix for Probes,and ddPCR reactions.

Data collection on QX600 droplet reader.