iAMP® Streptococcus pneumoniae Detection Kit
The iAMP® Streptococcus pneumoniae Detection Kit is an extraction-free real-time fluorescent isothermal PCR assay based on Atila’s proprietary isothermal amplification technology intended for qualitative detection of Streptococcus pneumoniae DNA in nasopharyngeal/oropharyngeal swabs, cerebrospinal fluid (CSF), whole blood or plasma samples from individuals with signs of Streptococcus pneumoniae infection.
Each kit includes 96 tests.
This product is for research use only (RUO).
iAMP® Streptococcus pneumoniae Detection Kit
Streptococcus pneumoniae is a major bacterial pathogen that causes a wide range of invasive and non-invasive diseases. It is the leading cause of community-acquired pneumonia and can also cause meningitis, bacteremia (bloodstream infection), and sinus or ear infections. These illnesses can develop quickly and are especially dangerous for young children, older adults, and individuals with weakened immune systems, often leading to hospitalization, long-term complications, or death if untreated. Because symptoms often overlap with other respiratory or bacterial infections, fast and accurate testing is essential to distinguish S. pneumoniae from other pathogens. Rapid and reliable testing enables faster treatment decisions, assists in preventing complications, and improves patient outcomes while reducing the spread of infection.
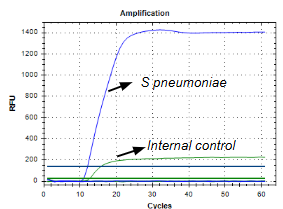
The iAMP® Streptococcus pneumoniae Detection Kit provides a simple, extraction-free workflow and delivers accurate results in under 90 minutes. Using Atila’s proprietary real-time isothermal PCR technology researchers can directly detect S. pneumoniae DNA from respiratory swabs, cerebrospinal fluid, whole blood, or plasma.
Features Include
Technical Specs
Workflow Overview for a Nasopharyngeal Swab.
This section is for demonstrative purposes only and may be incomplete or inaccurate. Always refer to the product instructions for precise guidelines and directions.
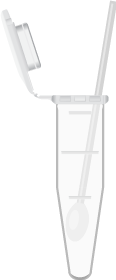
Dry swab in sample tube.

Add 750µL 1X sample buffer (1XSB) to dry swab.

Vortex briefly (2-3s) & incubate at 95°C for 10 minutes.
Add 10µL sample into Zebra BioDome tubes.

Run on standard PCR instrument.