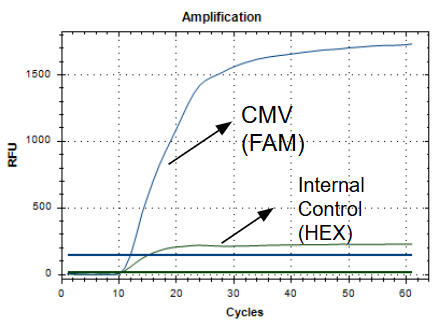
iAMP® cCMV Detection Kit
The iAMP® cCMV Detection Kit (congenital CMV) is an extraction-free real-time fluorescent isothermal PCR assay based on Atila’s proprietary isothermal amplification technology. This kit is intended for qualitative detection of DNA from human cytomegalovirus (HCMV), also called human herpesvirus 5 (HHV-5) in saliva and urine samples from neonates younger than 21 days of age.
Each kit includes 96 tests.
This product is for research use only (RUO).
iAMP® cCMV Detection Kit
Cytomegalovirus (CMV) is a ubiquitous virus that infects people worldwide and at high prevalence. Most infections are unnoticed and asymptomatic in children and adult populations. CMV transmission occurs through contact with body fluids from infected individuals. Although the majority of the infections are mild in nature, congenital infections of neonates and newborns can be serious. CMV is the leading cause of congenital infections worldwide and is the leading cause of nongenetic sensorineural hearing loss. Identification of congenital CMV infection within the first three weeks of life is highly beneficial in aiding healthcare providers in timely treatment and management of the infection and can lead to better hearing and cognitive outcomes.
Atila’s iAMP® cCMV Detection Kit features a simple, extraction-free workflow and delivers accurate results in under 90 minutes, making it a practical tool for early detection and preventing long-term complications. Timely diagnosis allows healthcare providers to begin appropriate treatment, reduce the risk of hearing loss, and monitor developmental milestones more effectively, helping to protect both the immediate and future health of affected infants.
Features Include
Fast Speed
Simple Protocol
Low Cost
Flexibility
Technical Specs
Reaction Time
Sample Types
Sensitivity
Instruments
Workflow Overview for the iAMP® cCMV Detection Kit for a Saliva Swab
This section is for demonstrative purposes only and may be incomplete or inaccurate. Always refer to the product instructions for precise guidelines and directions.
Workflow Overview for the iAMP® cCMV Detection Kit for a Urine Sample
This section is for demonstrative purposes only and may be incomplete or inaccurate. Always refer to the product instructions for precise guidelines and directions.