ScreenFire® HPV RS Kit (Zebra BioDome Format)
ScreenFire® HPV RS Kit is an extraction-free extended HPV genotyping assay, which identifies 13 specific high-risk HPV types in four groups beyond HPV 16 and 18 (HPV16, HPV18/45, HPV31/33/35/52/58 and HPV39/51/56/59/68), offers significant value in cervical cancer screening and management by improving risk stratification, guiding clinical decisions, and optimizing patient care.
- Extraction-Free
- Contamination Free
- Prepacked reagents
- Offers a portable and affordable reader
- Sample to result in an hour
- Perfect for mobile clinics
Each kit includes 96 tests.
This product is for research use only.
ScreenFire® HPV RS Kit (Zebra BioDome Format)
Human papillomaviruses (HPV) are small viruses containing a double-stranded, circular DNA genome of approximately 8000 base pairs. The viral genome contains early (E1-E7) and late genes (L1-L2), as well as a long control region (LCR). More than 200 different HPV types have been identified based on differences in genome sequences. Among these HPV types, a subset of 19 HPV types is considered as high/moderate high-risk for causing lesions of the cervical mucosa and leads to cervical cancer in women.
According to the World Health Organization (WHO), HPV is the second biggest cause of female cancer mortality worldwide, claiming about 250,000 lives annually. In Europe alone, the disease claims about 15,000 lives each year. There has been much evidence on the role of HPV genotype in cervical precursor lesions and invasive cancer. It is estimated that nearly 70% of cervical cancers are caused by HPV types 16 and 18.
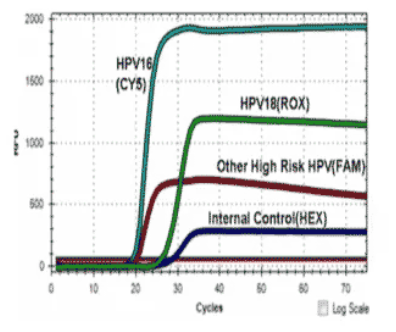
The ScreenFire HPV RS Kit is an isothermal nucleic acid amplification assay for the qualitative detection of high-risk types of human papillomavirus (HPV). High-risk HPV specific primers and fluorescent probes are used to amplify regions of viral genomic DNA including E6/E7 regions under isothermal conditions. The assay detects HPV types 16 alone, 18/45 as a group, 31/33/35/52/58 as a group (alpha-9 HPV group) and 39/51/56/59/68 (other high-risk HPV group) as a group.
Features Include
Technical Specs
Workflow Overview for the ScreenFire® HPV RS Kit (Zebra BioDome Format)
This section is for demonstrative purposes only and may be incomplete or inaccurate. Always refer to the product instructions for precise guidelines and directions.
Clinical dry swab in test tube.

Add 1mL 1XLB.

Vortex briefly (2-3s) and incubate 20 minutes at 95°C.
Add 10µL processed sample to well 1-8 of the strips.

Run on standard PCR instrument.