iAMP® H5N1 Detection Kit
iAMP® H5N1 Detection Kit is an extraction-free real-time fluorescent isothermal assay based on Atila’s proprietary isothermal amplification technology intended for the qualitative detection of H5N1 nucleic acids in nasopharyngeal, nasal, oropharyngeal, and/or conjunctival swab from individuals with signs and symptoms of H5N1 infection.
Each kit includes 96 tests.
- Fast speed: From sample to detection in less than 90 minutes
- Easy to Use: Simple, extraction-free workflow that saves time and money. The reagents are pre-loaded in the reaction tubes and sealed with gel. The only hands-on work is loading the samples.
- Flexibility: Our kits can be used for detection of any number of samples without wasting extra reagents.
- Low cost: Price per assay is much better than any other assay on the market for detection of H5N1.
This product is for research use only (RUO).
iAMP® H5N1 Detection Kit
An H5N1 infection is a serious illness caused by the H5N1 strain of the influenza A virus, commonly known as bird flu. While the virus primarily infects birds, it can occasionally spread to humans, typically through close or direct contact with infected birds, their droppings, or contaminated environments such as live bird markets. Human infections are rare but extremely severe, often presenting with high fever, cough, sore throat, muscle aches, and difficulty breathing. H5N1 is not currently easily transmitted between people, but health experts closely monitor it because of its potential to mutate and cause a global pandemic. Early detection, widespread testing, and ongoing surveillance are essential to prevent the spread of this highly pathogenic virus and to protect both public health and animal populations.
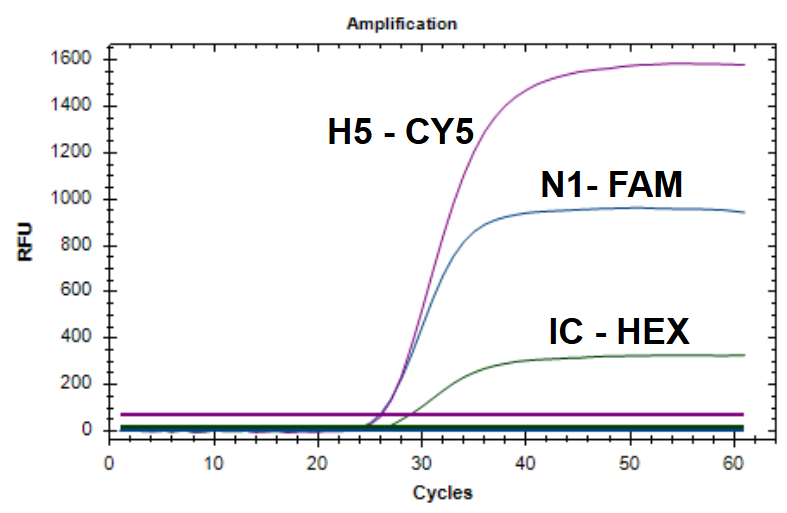
Atila’s iAMP H5N1 Detection Kit features a simple, extraction-free workflow and delivers accurate results in under 90 minutes, making it a practical tool for early detection and containment efforts. Early testing and surveillance remain critical in managing the risk posed by this highly pathogenic virus.
Features Include
Technical Specs
Workflow Overview for the iAMP® H5N1 Detection Kit
This section is for demonstrative purposes only and may be incomplete or inaccurate. Always refer to the product instructions for precise guidelines and directions.
Dry swab in sample tube.

Add 1mL of 1X Elution Solution (1XES) to dry swab.

Vortex briefly (2-3s) and incubate for 5min at room temperature to hydrate the swab.
Load 10µL of processed sample and quickly spin down.

Run on standard PCR instrument.