iAMP® EBV Detection Kit
The iAMP® EBV Detection Kit is an extraction-free, real-time fluorescent isothermal PCR assay based on Atila’s proprietary isothermal amplification technology intended for qualitative detection of human herpes virus 4 (HHV-4) also known as Ebstein-Bar Virus (EBV) DNA in nasopharyngeal/oropharyngeal swabs, cerebrospinal fluid (CSF), whole blood or plasma samples from individuals with signs of EBV infection.
This product is for research use only (RUO).
Each kit includes 100 tests.
iAMP® EBV Detection Kit
Epstein–Barr virus (EBV) is a member of the herpesvirus family and is a double-stranded, enveloped DNA virus (~172 kb). EBV infection in humans is extremely common, more than 90% of adults are infected, with the majority of cases the virus remaining in latent phase for life. While often asymptomatic, EBV can cause infectious mononucleosis in a subset of newly infected adolescents and adults. EBV infection is also associated with several cancers, including nasopharyngeal carcinoma, Burkitt lymphoma, and Hodgkin lymphoma. In individuals with congenital or acquired immunodeficiency, such as transplant recipients or patients with HIV/AIDS, EBV can lead to lymphoproliferative disorders.
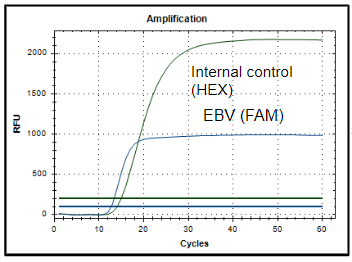
The iAMP® EBV Detection Kit provides a simple, extraction-free workflow and delivers accurate results in under 90 minutes. Using Atila’s proprietary real-time isothermal PCR technology researchers can directly detect EBV DNA from respiratory swabs, cerebrospinal fluid, whole blood, or plasma. Atila’s iAMP® EBV Detection kit can be used on variety of RT-qPCR instruments with 2 fluorescence channels.
Features Include
Fast Speed
Easy to Use
Low Cost
Flexibility
Technical Specs
Reaction Time
Sample Types
Sensitivity
Instruments
Workflow Overview for the iAMP® EBV Detection Kit (Nasopharyngeal swab)
This section is for demonstrative purposes only and may be incomplete or inaccurate. Always refer to the product instructions for precise guidelines and directions.
Workflow Overview for the iAMP® EBV Detection Kit(Plasma sample)
This section is for demonstrative purposes only and may be incomplete or inaccurate. Always refer to the product instructions for precise guidelines and directions.